Deconstructing the Infection Machinery of SARS-CoV-2
08/03/2021
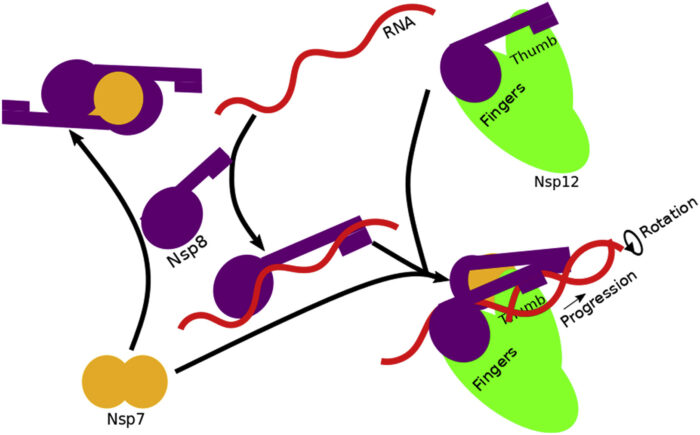
Possible assembly and mechanistic roles of components of the replication transcription complex (RTC) from SARS-CoV-2 responsible for recognizing and processing RNA. The solved crystal structure (top left) reported in this work exists in a dynamic equilibrium and forms at high concentrations of Nsp7 and Nap8. [Reprinted under a Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0/) from Wilamowski, et al. 2021. DOI: 10.1016/j.bpj.2021.06.006]
The Summary
The replication transcription complex (RTC) from the virus SARS-CoV-2 is responsible for recognizing and processing RNA for two principal purposes: copying viral RNA for propagation into new virus and ribosomal transcription of viral proteins.
Scientists conducted a systematic structural investigation of three components of the RTC—Nsp7, Nsp8, and Nsp12—and solved high-resolution crystal structures of the Nsp7/8 complex, providing insight into the interaction between the proteins.
The investigation involved a broad approach using a range of structural biology techniques, including small-angle x-ray and neutron solution scattering (SAXS and SANS) on all components and multiangle light scattering-coupled SAXS to identify which combination of components forms transient or stable complexes. Results indicated that individual Nsp7, Nsp8, and Nsp12 structures vary based on whether other proteins in their complex are present.
Combining the determinations of crystal structure, atomic coordinates reported elsewhere, SAXS, SANS, and other biophysical techniques, the multidisciplinary research team provided insight into RTC assembly, mechanism, and potential avenues for disruption of the complex and its functions.
Related Links
- BER Resource: Center for Structural Molecular Biology
- BER Resource: Structural Biology Center
- BER Resource: Structurally Integrated Biology for the Life Sciences
- Feature Story: Deconstructing the Infectious Machinery of SARS-CoV-2
- News Release: Deconstructing the Infectious Machinery of the SARS-CoV-2 Virus
References
Mateusz Wilamowski, Michal Hammel, Wellington Leite, Qiu Zhang, Youngchang Kim, Kevin L. Weiss, Robert Jedrzejczak, Daniel J. Rosenberg, Yichong Fan, Jacek Wower, Jan C. Bierma, Altaf H. Sarker, Susan E. Tsutakawa, Sai Venkatesh Pingali, Hugh M. O’Neill, Andrzej Joachimiak, Greg L. Hura, “Transient and stabilized complexes of Nsp7, Nsp8, and Nsp12 in SARS-CoV-2 replication,” Biophys. J. 120 (15), 3152-3165 (2021). [DOI: 10.1016/j.bpj.2021.06.006]
